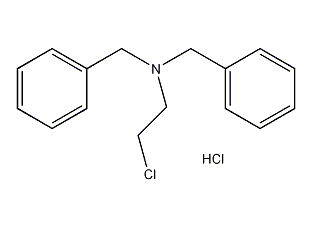
structural formula
| business number | 016u |
|---|---|
| molecular formula | c16h18cln·hcl |
| molecular weight | 296.23 |
| label |
dibenzylaminoethane chloride hydrochloride |
numbering system
cas number:55-43-6
mdl number:mfcd00012518
einecs number:200-234-5
rtecs number:hq6825000
brn number:none
pubchem number:24857529
physical property data
1. character:white crystalline powder.
2. density (g/ml,25/4℃): 1.250
3. relative vapor density (g/ml,air=1): undetermined
4. melting point (ºc): span>192
5. boiling point (ºc,normal pressure): undetermined
6. boiling point (ºc,5.2kpa): undetermined
7. refractive index: undetermined
8. flashpoint (ºc): undetermined
9. specific optical rotation (º): undetermined
10. autoignition point or ignition temperature (ºc): undetermined
11. vapor pressure (kpa,25ºc): undetermined
12. saturated vapor pressure (kpa,60ºc): undetermined
13. heat of combustion (kj/mol): undetermined
14. critical temperature (ºc): undetermined
15. critical pressure (kpa): undetermined
16. oil and water (octanol/log value of water) partition coefficient: undetermined
17. explosion limit (%,v/v): undetermined
18. lower explosion limit (%,v/v): undetermined
19. solubility:stable in acidic solutions, but rapidly destroyed in neutral or alkaline solutions. soluble in dilute acid, 95%ethanol and propylene glycol are almost insoluble in water.
toxicological data
1, acute toxicity: mouse abdominal cavity ld50: 75mg/kg; mouse subcutaneous ld50: 400mg/kg; mouse intravenousld50: 50mg/kg; feline vein ldlo: 35mg/kg
ecological data
none
molecular structure data
5. molecular property data:
1. molar refractive index:78.12
2. molar volume (m3/mol):234.6
3. isotonic specific volume (90.2k):599.7
4. surface tension (dyne/cm):42.7
5. polarizability(10-24cm3): 30.97
compute chemical data
1. reference value for hydrophobic parameter calculation (xlogp): none
2. number of hydrogen bond donors: 1
3. number of hydrogen bond acceptors: 1
4. number of rotatable chemical bonds: 6
5. number of tautomers: none
6. topological molecule polar surface area 3.2
7. number of heavy atoms: 19
8. surface charge: 0
9. complexity: 187
10. number of isotope atoms: 0
11. determine the number of atomic stereocenters: 0
12. uncertain number of atomic stereocenters: 0
13. determine the number of chemical bond stereocenters: 0
14.nodetermine the number of stereocenters of chemical bonds: 0
15. the number of covalent bond units: 2
properties and stability
none
storage method
this product should be sealed and stored in a cool place.
synthesis method
none
purpose
for biochemical research. medicine.
extended-reading:https://www.bdmaee.net/wp-content/uploads/2022/08/37-5.jpgextended-reading:https://www.bdmaee.net/tris3-dimethylaminopropylamine-2/extended-reading:https://www.bdmaee.net/toyocat-dmch-hard-bubble-catalyst-for-tertiary-amine-/extended-reading:https://www.newtopchem.com/archives/820extended-reading:https://www.bdmaee.net/wp-content/uploads/2022/08/-t-12-tin-catalyst-nt-cat-t-120–t-12.pdfextended-reading:https://www.newtopchem.com/archives/40275extended-reading:https://www.cyclohexylamine.net/dabco-ne1060-non-emissive-polyurethane-catalyst/extended-reading:https://www.bdmaee.net/fascat2004-catalyst-cas7772-99-8-stannous-chloride/extended-reading:https://www.newtopchem.com/archives/1880extended-reading:https://www.bdmaee.net/dabco-ncm-catalyst-cas110-18-9–germany/

